Brown Dogs and Red Herrings
Or, why we no longer experiment much on dogs.
On a balmy June morning in 2007, a pair of Pennsylvania congressmen gathered their colleagues in the basement of the Rayburn House Office Building to show them a video. The lawmakers watched excerpts from a documentary that had aired on HBO the previous winter, about an unscrupulous dog dealer in Arkansas who was selling animals to laboratories. About 40 years after Pepper's story prompted the first federal dog-napping law, the wheels were in motion on another. Mike Doyle, D-Pa., introduced the film: "It's hard to believe that someone would steal people's pets and treat them this way."
Despite the film, the Pet Safety and Protection Act failed to pass in 2007, just as it has on a nearly annual basis for the last 13 years. Should it ever succeed, the law would ban the sale of any dogs and cats for research by "Class B" dealers—those who collect or buy their animals rather than breeding them themselves. (If such a designation had existed in 1965, it would have applied to the three men—Jack Clark, Russ Hutton, and Bill Miller—who handled Pepper during her 10-day journey to the dog lab.) That's not an unreasonable goal—as the HBO special showed, the Class B dealers are almost certainly engaging in some shady practices—but it hardly seems worth the sustained efforts of the nation's top animal welfare advocates. To put things in perspective, only about a dozen of these dealers remain in business, and they sold fewer than 3,000 dogs to research labs in 2008. Those dogs, stolen or not, represent just afew thousandths of 1 percent of all the animals used by American scientists.
It's not too long ago that dogs and cats mattered a great deal to the practice of biomedical research. Today they're barely relevant—a tiny, shrinking line item on an enormous ledger of death. Something like 100 million animals are killed in experiments every year in the United States, yet groups like the Humane Society of the United States and the Animal Welfare Institute continue to treat a handful of pet thefts as a keystone issue. How did a broad struggle for the ethical treatment of lab animals turn into a border skirmish over missing pets?

Dogs were already a favorite of scientists when live-animal experimentation became widespread in the mid-19th century. Bred for docility over many thousands of years, the dog evolved as an ideal animal for the lab, simple to feed and house and poke and slice. It was also easy enough to imagine man's best friend as a stand-in (and a physiological model) for people: What's true for Pepper must be true for us; her heart must beat like mine.
Potential test subjects were everywhere in the early days of vivisection. A fascinating paper by historian Bert Hansen describes how American cities of the Gilded Age were beset with stray and wild dogs. In 1885, the New York Herald guessed there were some 300,000 in Manhattan and another 150,000 in neighboring Brooklyn. The scourge invited open brutality: Police officers were empowered to shoot dogs in the midst of rabies panics, and captured strays were crammed into cages and drowned in the East River.
From the start, though, the use of dogs for violent laboratory experiments disturbed a bourgeois sensibility that associated them—or at least the purebreds—with family and the comforts of domestic life. (Many years later, Julia Lakavage would tell reporters that Pepper and her other dogs were "like family members … children that don't grow up.") That notion extended to the very upper reaches of society: Queen Victoria of England—whose menagerie of pets included Skye terriers Islay, Cairnach, and Dandie Dinmot; King Charles spaniel Dash; greyhound Nero; mastiff Hector; and many more—was well-known to be suspicious of the new science of animal experimentation. In 1875, the year before the passage of Britain's Cruelty to Animals Act, Victoria asked Joseph Lister to speak out against vivisection: "The Queen has been dreadfully shocked at the details of some of these practices," her secretary wrote, "and is most anxious to put a stop to them."

By the first decade of the 20th century, the plight of research dogs had become the principal cause for animal activists, who singled them out on account of their vulnerable nature—and the ease with which they could be used to elicit public support. In February 1903, a pair of Swedish animal lovers slipped into a medical demonstration at University College London and watched physiologist William Bayliss cut open the neck of a brown dog. In their published account of the episode, they claimed that the animal had suffered under improper anesthesia. Riots erupted after a monument was erected to the anonymous brown dog. (Tensions over the "brown dog affair" have been revived in England several times since.)
Scientists responded to the furor over experimental dogs with anger and derision. Historian Susan Lederer cites a 1910 screed by a New York physician who accused the anti-vivisectionists of suffering from a kind of "zoophilic psychosis": They "interest themselves not so much in experiments upon fishes, insects, pigeons, rats, mice, snakes, nor in cruelties to men, cattle, chickens, and sheep," he wrote. "Their interests are bent towards those useless animals which can be made the objects of fondling and which compared with other animals play a minor role in the great field of scientific experimentation."
Within a few years, the concern over experimental dogs crystallized into a fear that pets were being stolen and sold to research institutions. The specter of "dog-napping" became central to the activist rhetoric starting in the 1910s, says Lederer, and soon showed up in animal-welfare exhibitions and pamphlets with titles like Guard Your Dog. Meanwhile, the dog-theft meme percolated through the media—and not only in the newspapers of ardent anti-vivisectionist William Randolph Hearst. In the early 1920s, the New York Times offered regular coverage of a gang of "auto-pirates" who pilfered more than 150 animals from wealthy estates on Long Island. That story, and others like it, were summed up in a Times headline from February 1922: "Stealing Fine Dogs a Regular Industry … Most Intelligent of Their Kind, but Have Never Learned How Not To Be Stolen."
The epidemic of pet theft received intermittent coverage over the next few decades. With the re-emergence of the animal welfare movement in the 1960s—and the disappearance of Pepper in Pennsylvania—it became a national story.

At the turn of the century, animal activists organized their campaigns around research dogs and cats, but science was already moving in a new direction. Mendel's ideas had been rediscovered in 1900 and the term genetics coined in 1906; the new science of heredity created a sudden need for organisms whose exact ancestry was known and controlled. Within a few years, the perception and practice of biomedical research had begun to diverge: While humane societies were worrying about missing pets, researchers were shifting to a new set of standardized, purpose-bred laboratory organisms—the rat, the mouse and the fruit fly.
The standardization of laboratory animals reflected the growing influence of industrial engineering in America. Under the new paradigm, research organisms could be seen as a sort of raw material for use in a knowledge assembly line. Just 10 or 20 years earlier, a physiologist might have conducted his experiments on a variety of species—frogs and rabbits, for example, or dogs, horses, and baboons—to show that a finding could be generalized across the whole of creation. Now, whatever could be demonstrated in a rat or a mouse was assumed to be true of a dog, a horse, or a human. The growth of statistical science also changed the nature of laboratory work: Now it was advantageous to increase your N by repeating an experiment as many times as you could on as many animals as were available.
Biomedical science became even more industrialized during the postwar period as government money flowed through the pumped-up National Institutes of Health. Grants officers realized that animals could be most efficiently distributed if they were churned out in large numbers at a few, centralized locations. Meanwhile, the organisms themselves had begun to be packaged as commercial products: In 1941, the breeders at the Jackson Laboratory in Maine received a patent on their line of "JAX Mice" research animals; a year later, the Wistar Institute in Philadelphia trademarked its own line of "WISTARATs."
Experimental dogs had been well-suited to the old-fashioned, artisanal mode of inquiry—William Harvey cutting open a heart, Christopher Wren pumping a vein full of wine, Richard Lower transfusing blood with a chain of quills. But as an instrument of modern science, they were starting to look obsolete. The economics of breeding dogs were one problem: In 1965, the year Pepper was taken, a dog could be raised and maintained in a research lab for about 70 cents per day. At that rate, though, it would cost more than $250 to produce a single, sexually mature animal. A stray with uncertain heritage could be had from a dealer for just $15, which explains why hospitals were buying dogs like Pepper to begin with.
That's not to say dogs didn't have their niche in biomedicine. Medical schools, in particular, made ample use of them for education and research in surgery and cardiology. And many of the landmark studies on anemia, diabetes, hemophilia, and cardiothoracic surgery used dogs. But by the time of Pepper's death, there was already a move toward replacing canines on the operating table with a brand-new model organism—the miniature pig. The hairless and docile Yucatan mini-pig had been introduced to the United States in 1960, and the University of Missouri opened a dedicated miniature swine production facility in 1965.
Like dogs, miniature pigs were big enough to operate on but not so big that they couldn't be housed and handled. Porcine skin turned out to be so similar to our own that it could be used as a temporary patch for burn patients and a vehicle for the study of melanoma. Mini-pigs were also excellent subjects for the study of cardiovascular fitness, atherosclerosis, obesity, and diabetes.
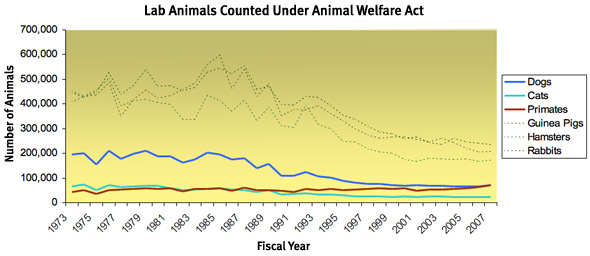
A total of 260,000 dogs were used by U.S. labs in 1967, the year that Pepper's dog-napping law took effect. Over the next four decades, that number would drop by almost 75 percent. Surely some of that decline has come in response to continuing pressure from animal-welfare activists. (Research on miniature pigs isn't as likely to draw angry protests.) More significant, though, was the fact that science itself had changed.

The Laboratory Animal Welfare Act passed in 1966 gave some protection to a small number of species in limited settings. But the animal rights activists, emboldened by success, were soon able to broaden its scope. The first major amendments arrived in 1970; from then on, the law would be known simply as the "Animal Welfare Act," and its reach extended to circuses, zoos, shows, and wholesale pet dealers. The laboratories, too, had new responsibilities: Where the 1966 version had applied only to research holding facilities—with cage-size and feeding requirements, for example—now the experiments themselves were subject to humane standards, including pain management whenever possible. But what might have been the most significant change to Pepper's law was a broadening of the very definition of the word animal. Instead of protecting only the most lovable critters—dogs, cats, nonhuman primates, rabbits, hamsters, and guinea pigs—the new law could be taken to apply to every other warm-blooded animal used in the lab.
That dramatic mandate was handed over to Dale Schwindaman, a federal veterinarian who had been summoned to Washington four years earlier to help draft the original regulations related to Pepper's law. It hadn't been easy for the Department of Agriculture to enforce the original act; now Schwindaman had to figure out how to address a raft of new responsibilities.
The numbers were grim. A laboratory-animal survey conducted by the National Academies of Science found that the original covered species—the cute animals—made up just 7 percent of the total used in U.S. labs. In addition to the 260,000 dogs, there were 100,000 cats and 800,000 hamsters. But there were 30 million rats and mice. "The job was too big," Schwindaman says. Full coverage of warm-blooded species would have given the USDA the obligation to supervise every last junior college that used rodents or chick embryos for teaching. "The math just did not allow it. … We couldn't use the National Guard to make all of these inspections. We didn't have the force."
With Schwindaman's help, the USDA put in place in 1972 a special exemption for rats, mice, and birds, allowing scientists to treat them however they saw fit—in cages of any size, in experiments with any degree of pain and suffering. That exemption remains in force, despite Schwindaman's later attempts to overturn it. To this day, 95 percent of the animals used in research labs receive no federal protection whatsoever under the Animal Welfare Act.

In the fall of 2001, an undercover animal activist took a job cleaning rat and mouse cages on the Chapel Hill campus of the University of North Carolina. Over the next six months, she would collect more than 40 hours of footage on behalf of People for the Ethical Treatment of Animals with a camera hidden under her lab coat. The video showed researchers marking newborn mice by amputating their toes and cutting the brains from baby rats without anesthesia. Rodents were trampled to death in overcrowded cages, left to die in garbage bins, or allowed to suffer with swollen tumors and open sores.
The USDA had just agreed, in 2000, to extend laboratory-animal protections to rodents and birds. (The decision, by Agriculture Secretary Daniel Glickman, came out of a legal settlement with animal rights groups, which had sued the government over its arbitrary redefinition of the term animal almost 30 years before.) By the spring of 2002, PETA was already fighting to keep the old rule from coming back. Sen. Jesse Helms had added a line to that year's Farm Bill that would make the exclusion of purpose-bred rats, mice, and birds a matter of federal law. ("A rodent could do a lot worse than live out its life span in research facilities," he said.) The footage from Chapel Hill—which PETA sent to every member of the conference committee—did little to change the debate: When Congress reached a final agreement on the Farm Bill a week later, the Helms amendment was still in place. Protection for rats, mice, and birds had once again been stripped from the Animal Welfare Act.
"That was such an enormous disappointment," says PETA's director of laboratory investigations, Kathy Guillermo. The story of a single dog had once been enough to pass the country's most important animal-welfare law, but evidence of repeated rodent abuse in a government-funded lab barely made the news. "It's because it's mice and rats," Guillermo says. How do you protect an animal that most Americans would be happy to kill themselves with glue traps, even, or poison?

The rodents' PR problem helps explain how they came to infest science and medicine in the first place. C.C. Little, the inventor of the modern lab mouse and founder of the Jackson Laboratory, had long used "the age old enmity of woman and the Muridae" as a sales pitch for his model organism. Writing in American Naturalist in 1939, he noted that "it has been difficult to keep at fever heat a sufficient level of sympathy for the rodent similar to that which the dog or cat engenders." Indeed, when controversy erupted over Pepper's death in 1965, Little's company was quietly supplying physiologists with close to 1 million mice every year. (Today, the sales of rats and mice dominate a $1.4 billion global market in laboratory animals.)
For 100 years or more, scientists and activists had traded blows over the ethics and practice of research on dogs and cats. Through all that back-and-forth, lab rodents were always left just across the moral frontier of live-animal experimentation—close enough to humans to remain a meaningful source of knowledge but not so close that we couldn't slaughter them in droves. Yet it's not obvious—to those who might consider the question—that the welfare of a rat or mouse is any less important than that of a dog. Recent research suggests that the health of mice improves when they're given cage toys, running wheels, and crawl tubes to play with. Rats can learn to respond to a name and recognize individual people. We might quarrel over the inner lives of honeybees or river trout, but is the suffering of our fellow mammals really in question?
Meanwhile, rats and mice are subject to some of the most extreme and invasive experiments in biomedicine. By the early 1980s, we were spiking mouse DNA with cancer-causing genes; a few years later, we started to "knock out" specific lines of genetic code. (Scientists mapped out the entire mouse genome in 2002 and the rat genome in 2004.) We regularly subject rodents to pain, starvation, solitary confinement, and grotesque disfigurement. Whatever misery they endure is multiplied across the hundreds of millions of rats and mice used in labs every year.
The animal-welfare groups have failed in their most ambitious efforts to protect laboratory rodents. "We did and do strongly support the inclusion of rats and mice," says Cathy Liss, current president of the Animal Welfare Institute. "But the question is how can we properly address that? At this juncture, it's premature to go forward and rally support." With rodents off the table, though, it's not clear what's left for the activists to do.
