
Science Fiction Becomes Science-Fact: Two Strategies for Repairing Humans
New breakthroughs in the field of regenerative medicine are allowing us to live longer, healthier lives.
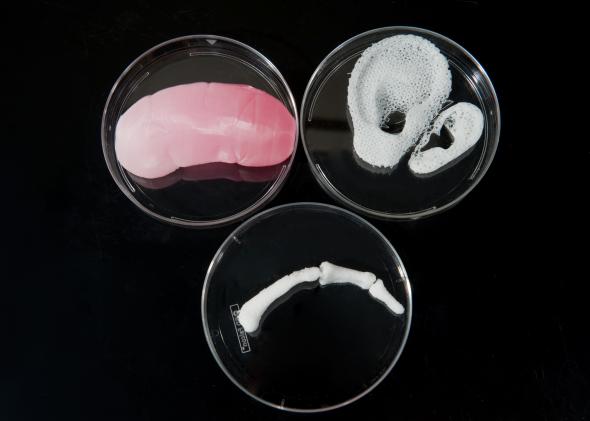
Photo Courtesy Wake Forest Institute for Regeneratie Medicine.
Not long ago, it would have sounded like science fiction to discuss growing human organs in the lab or re-writing DNA. Yet today both are realities that will change the world and allow for longer and healthier lives.
Already, lab-grown bladders, windpipes and blood vessels have been successfully created and implanted into humans. Most recently, tissue engineering pioneer Dr. Anthony Atala and his team at the Wake Forest Institute for Regenerative Medicine announced another breakthrough: lab-made vaginas—one of the most complex organs made to date. In four girls with MRKH syndrome, a medical condition in which the vagina and uterus are underdeveloped or absent, Dr. Atala’s team was able to create new organs that functioned normally, dramatically increasing each patient’s quality of life.
To grow the organs, the scientists created scaffolds in the proper size and shape and lined the structures with each patient’s vaginal epithelial cells on one side and their muscle cells on the other. The structure was then placed in an incubator for about a week before being implanted into each patient. The vagina is harder to grow than other organs because it highly elastic and secretes mucus.
It has now been more than five to eight years since each of the girls received the transplants and all patients have normally functioning organs. This durability of lab-grown organs shows that, today, regenerative medicine is not simply offering petri dish hope, but real solutions and real cures.
“I feel fortunate because I have a normal life, completely normal,” said one of the patients.
Although there have been many successes to date, the days of organ-growing are still early. Dr. Atala’s Institute is currently working on engineering more than 30 different replacement tissues and organs with “the goal to cure, rather than merely treat, disease.” And there are researchers all over the world who are similarly focused.
On the same day that Dr. Atala’s paper was published, a group of scientists from Switzerland and the United Kingdom published data showing that they had successfully created lab-grown nostrils to replace cancer-ravaged noses in two women and three men. The day will come when hearts, lungs, and livers are available for replacement or repair – the question is not if, but when and how.
Creating organs out of scaffolds is not the only way to build them. Some scientists are experimenting with 3D printers that print cells on to bio-paper. Others, like genome pioneer Dr. Craig Venter, are attempting to rewrite the DNA for pig organs so that they can be used in humans without rejection.
It’s a little bit like a hardware vs. software approach. Dr. Atala’s lab-grown organs won’t be rejected because he uses the patient’s own cells. Dr. Venter, in his typical maverick style, is looking to rewrite the pig’s source code so that their organs can easily be swapped for human parts.
“We're going to start with generating a brand new super-accurate sequence of the pig genome, and then go through in detail and compare it to the human genome,” said Dr. Venter. “The goal is to go in and edit, and where necessary, rewrite using our synthetic genomic tools, the pig genes that seem to be associated with immune responses.”
Over a decade ago, Dr. Venter and his team mapped the first human genome in tandem with the U.S. government’s Human Genome Project, and in 2010 they created the first synthetic life form. The next logical step after reading and writing code is to begin to edit. Bioengineers all over the world are excited about this prospect and even traditional software companies are looking to participate.
For instance, software design company Autodesk recently announced that it created and 3D-printed a synthetic virus for a mere $1000, a big accomplishment that built upon the knowledge generated by Dr. Venter’s earlier (and more expensive) success.
Whichever method of regenerative medicine reaches the public first, there is no question that it is necessary. Today there are over 120,000 people on the US organ waitlist. Around the world, the global organ shortage affects millions. And, according to Dr. Venter’s Synthetic Genomics, “In the United States alone, about 400,000 people die annually from various forms of lung disease including cancer, but scarcely 2,000 people are saved with a lung transplant and only about 2,000 are added to the transplant wait list annually.” It is about time for science fiction to turn into science fact.