A big asteroid impact is one of nature’s more terrifying events. A chunk of rock and metal comes screaming out of the sky, slamming into the ground 20 times faster than a rifle bullet, exploding with the energy of a multimegaton nuclear bomb, creating a blast wave that incinerates everything for kilometers around.
But they’re not all bad. They make diamonds.
Yes, seriously. The idea is that the huge pressures and heat generated when the asteroid hits the ground are so powerful they can make diamonds out of carbon. Some evidence of this has been found, but until now the science has been shaky. A team of scientists from around the globe has just published new results, though, showing that diamonds can indeed be formed in just this way, and let me be honest: There is nothing about this story that isn’t totally cool. Lasers! X-rays! Asteroid impacts! Precious gems!
So yes, first, let’s talk diamonds for a minute. You may already know diamonds are a form of carbon. When you think of carbon you probably picture a soft, black material like coal. That’s amorphous carbon, where the atoms are connected together all willy-nilly. There’s no real overall structure to the substance.

Shutterstock/nobeastsofierce
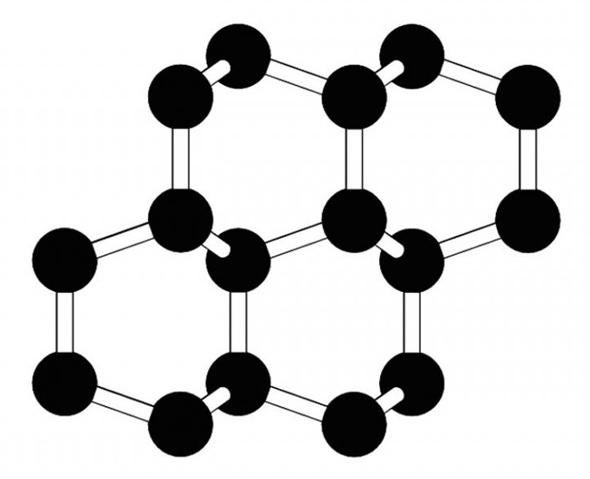
Graphite is another form of carbon, where the atoms bond together to form interconnected hexagons in flat sheets (different structures using the same elements are called allotropes). Graphite is surprisingly strong along the plane of each individual sheet, but the sheets themselves slide around on top of each other easily.
Shutterstock/Vasilyev
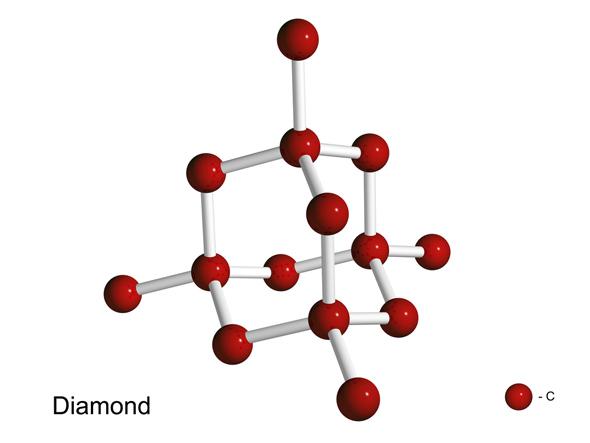
Diamonds are a third form of carbon, where the atoms come together to bond in the form of interconnected tetrahedra (like the corners of a four-sided triangular pyramid). A diamond is essentially a gigantic carbon molecule, and the bonds are strong in all three dimensions. That’s why diamonds are so hard; the bonds are very tough to break. Terrestrial diamonds form when carbon under the Earth’s crust is subjected to high pressure and temperature, allowing the atoms to assemble in the tetrahedral pattern.
Diamonds come in different forms, too. One is called lonsdaleite, named after crystallographer Kathleen Lonsdale. It has a slightly different structure, with the tetrahedra connected in a zig-zaggy hexagonal shape; this was shown back in 2014. Lonsdaleite is not thought to occur inside the Earth naturally. It’s been found in meteorites, though, like Canyon Diablo—the remnants of a nickel-iron asteroid that slammed into the Arizona desert 50,000 or so years ago.
Péter Németh
Well, maybe. The diamond crystals found in the meteorites aren’t pure, and the identification of them has been controversial. Scientists have tried to create lonsdaleite in the lab in the past but haven’t been totally successful.
Until now. This is the second incredible part of this story. And it has LASERS!
To attempt the creation of lonsdaleite, the researchers fired two high-powered lasers into a sample of graphite. By high power, I mean gigawatt lasers, which is a lot. They only fire them in pulses lasting 10 nanoseconds, but still. That graphite sample knows who’s boss after that.
The huge energy dumped into the sample creates a shock wave, delivering a pressure of up to 2 million atmospheres, 2 million times the pressure of air at sea level. That’s enough to blast away the bonds between carbon atoms and let them rearrange themselves into the diamond-defining tetrahedra.
But how to prove it? Well, by doing X-ray crystallography, of course!
And here’s the third cool part. Crystals have well-defined structures. If you send a beam of X-rays through them, the photons diffract, bending around the structure. If you put an X-ray detector on the other side, you can measure the pattern generated as the photons bend. That pattern is unique for every crystal, like a fingerprint, allowing you to determine what kind of crystal you’ve made.
So the researchers sent a beam of X-rays through the graphite samples as the high-powered lasers were firing, allowing them to keep track of what was going on inside. This part slays me: The X-rays were sent in a 50 femtosecond pulse—a femtosecond is one-quadrillionth of a second, or 10-15 seconds. Imagine a microsecond, a millionth of a second. That’s a tiny amount, right? Now divide it into a billion slices. That’s a femtosecond.
The fact that we can manipulate matter and energy on scales this small makes the hair on the back of my neck stand up. In a good way. That’s simply amazing.
Anyway, they timed the X-rays to look at the graphite samples just as or just after the shock wave passed through the graphite sample. What they found was definitive evidence that they had created lonsdaleite!

Chris Setter
Moreover, they found that the graphite started turning into diamonds at a pressure of about half a million atmospheres, and pure lonsdaleite formed at 2 million. These are the kinds of pressures you see in asteroid impacts, like Canyon Diablo (which was probably a 30–50 meter wide chunk of iron).
I think it’s fantastic that this has finally been reproduced in the lab. The more we know about asteroid impacts, the better! Also, lonsdaleite’s structure likely makes it even stronger than regular diamonds, which has a lot of industrial applications. The experiment I’ve described here doesn’t produce very much of the mineral (the graphite samples used were about the same thickness as a human hair), but if the process can be made simpler and scaled up, then manufacturing lonsdaleite could become a reality.
I’m all for that, but really, it’s the science and engineering here that gets me excited. The things we can do, just to test nature and see how much of it we understand!
And sometimes we get sparkly gems out of it, too. Seems fair to me.
Tip o’ the tiara to Ron Baalke.
