The comet Hartley 2 has come and gone, and the NASA mission EPOXI is also moving on after an exceedingly close flyby of the comet’s solid nucleus. The pictures we got were fantastic and beautiful… but their real power comes from coupling them with spectra.
In the picture above – an enhanced version of one of the images taken during the space probe’s flyby – you can see fan-like emission coming from the comet’s nucleus. These are jets; sprays of material coming out of the nucleus. Comets are made of rock and ice, and when the comet nears the Sun, the heat can turn that ice directly from a solid to a gas. This gas then shoots out from pockets on the nucleus, creating these jets. The EPOXI team (including my old boss, Don Lindler!) made a fantastic animation from a series of observations showing these jets in action.
But what are these ices made of? Lots of things we normally think of as liquids or gas (water, ammonia, carbon dioxide, and so on) exist in comets. In many comets, we see lots of water, and in fact the Swedish satellite Odin detected about 200 kg/sec (440 pounds per sec) of water coming off Hartley 2! So is water powering these jets?
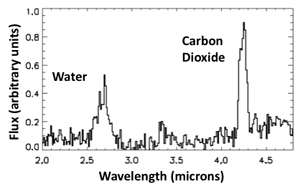 We can find out… using spectra. By breaking up the light from an object into its component colors, we can learn all manners of things including what it’s made of. EPOXI did just that with the jets streaming from Hartley 2, and while it did find water, amazingly, it found a lot more carbon dioxide!
We can find out… using spectra. By breaking up the light from an object into its component colors, we can learn all manners of things including what it’s made of. EPOXI did just that with the jets streaming from Hartley 2, and while it did find water, amazingly, it found a lot more carbon dioxide!
The graph there shows an infrared spectrum of the comet’s jets, and the emission from water and CO2 are labeled. Apparently, it’s pockets of dry ice (frozen CO2) that are powering these jets, with water taking something of a back seat. That CO2 has probably been frozen deep inside the comet for billions of years, and is just now seeing sunlight for the first time. And when it does, it flashes into a gas, pouring forth from the comet. It takes other things with it, too, including dust grains, water, and even organic compounds like methanol. These gases then surround the nucleus, forming the fuzzy head of the comet, and stream away to form its tail.
And we couldn’t know this unless we went to the comet to see for ourselves. Observations like this are very difficult or impossible to do from the ground, where the Earth’s atmosphere absorbs the comet’s faint infrared signature (it’s the very presence of water and CO2 in our air that absorbs the light from those molecules in the comet!). Sometimes, there’s no substitute for being there.
And studying comets is important. For scientific reasons, they represent pristine example of the early solar system. Locked up inside them are chemical compounds and mixtures probably unchanged since they formed billions of years ago. Comets are the time capsules of the solar system.
They’re important for selfish reasons, too. Hartley 2 passed us at the relatively safe distance of 18 million km (11 million miles); close by in astronomical terms, but far on a human scale. Not all comets pass us safely, though, and we know they sometimes impact the Earth. A comet like Hartley 2 has a nucleus about 2 km across, small as comets go. Still, an impact by an object that size would cause a global catastrophe… and bigger comets exist. The more we know about these objects the more we can figure out what to do if we see one that decides to pay us a closer call than Hartley 2.
Related posts:
- 3D House of Comet Nucleus!
- Amazing close-ups of comet Hartley 2!
- Arecibo gives comes some radar love
- Three views to a comet